Disordered N-terminal region of CLK1 oligomerizes for recruitment to nuclear substructures and splicing function
Disordered N-terminal region of CLK1 oligomerizes for recruitment to nuclear substructures and splicing function
George, A.; Fattet, L.; Capraro, D.; Jennings, P. A.; Adams, J.
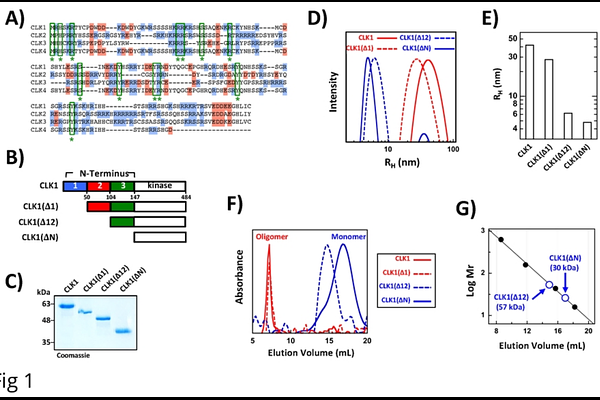
AbstractCdc2-like kinase 1 (CLK1) phosphorylates the serine-arginine (SR) proteins, a family of nuclear factors essential for mRNA splicing. The ability of CLK1 to recognize and efficiently modify SR proteins is strictly dependent on a lengthy, disordered N-terminus flanking its kinase domain. In addition to stimulating phosphorylation, this 150-residue extension also induces large oligomer formation in CLK1 but it is unclear whether said structure is important for catalytic or cellular function. We identified a subset of N-terminal residues that, upon removal, impairs oligomerization of CLK1 but does not abolish phosphorylation of the SR protein SRSF1. Despite robust phosphorylation, CLK1 lacking these high-order oligomerization sequences cannot effectively enter nuclear speckles and release SRSF1. This inability to mobilize CLK1 has detrimental effects on the alternative splicing of the CLK1 gene, severing an important, autoregulatory mechanism that controls active cellular levels of the kinase. Such findings indicate that whereas a limited group of residues in the N-terminus activates the kinase domain for SR protein phosphorylation, sequences that induce oligomerization direct CLK1 to the proper subnuclear structures for splicing function.