Photopharmacology in Action: Conformational Landscape of a Photoswitchable Covalent Kinase Inhibitor
Photopharmacology in Action: Conformational Landscape of a Photoswitchable Covalent Kinase Inhibitor
Duve, T.; Thallmair, S.
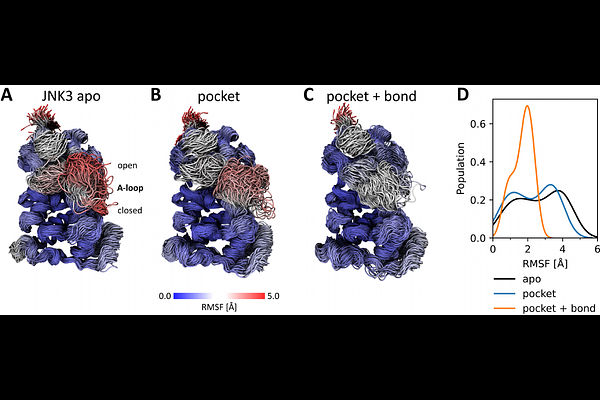
AbstractPhotopharmacology is a rapidly evolving field that uses light to control drug activity with high spatial and temporal precision, offering innovative therapeutic strategies with reduced side effects. In this study, we investigate a photoswitchable covalent inhibitor for the MAP kinase JNK3, a target for the treatment of neurodegenerative diseases. The inhibitor, which is based on a diazocine photoswitch, can undergo reversible photoisomerization of a double bond, switching between two (meta-)stable isomers. Atomistic molecular dynamics simulations, comprising over 35 s of total simulation time, reveal how the distinct conformational spaces of the two isomers modulate their interactions with JNK3 within the ATP-binding pocket. We show that only the metastable trans isomer can form a covalent bond with JNK3, thereby permanently inhibiting its function. In contrast, for the cis state the distance between the inhibitor and the targeted cysteine residue is too large to allow covalent bond formation. Furthermore, our simulations reveal that the covalent bond, in combination with the environment of the protein pocket, hinders the full back-relaxation of the trans isomer to the stable cis isomer. Finally, our data shows that the covalently bound stable cis isomer modulates the conformations of JNK3, mainly of its activation loop. Our findings provide molecular insights into the complex dynamics of photoswitchable inhibitors, guiding future drug design for light-controlled therapeutics.