Plastin-3 membrane recruitment drives cell-in-cell invasion during entosis
Plastin-3 membrane recruitment drives cell-in-cell invasion during entosis
Prapa, S.; Bozkurt, E.; Gandia, C.; Hernandez Cano, A.; Soriano, M.; Noon, L. A.; Gomez Sanchez, J. A.; Orzaez Calatayud, M.; Lucantoni, F.
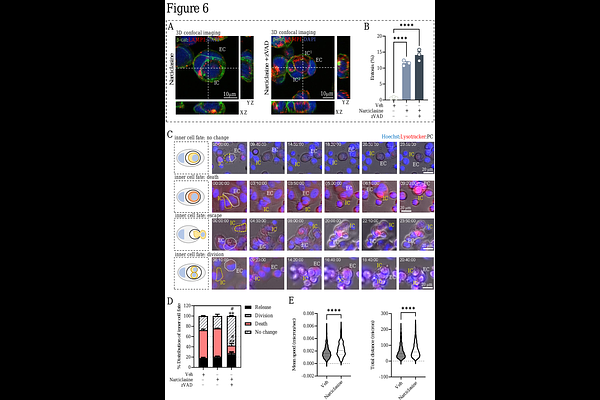
AbstractEntosis is a cell-in-cell (CIC) invasion process in which one living cell actively invades into another, with important implications for tumor evolution, cell competition, and responses to cellular stress. Both the initiation and progression of entosis require extensive actin cytoskeletal remodeling, as one cell must physically accommodate the complete internalization of another viable cell, which can subsequently migrate, divide, undergo degradation, or escape. Nearly two decades after its discovery, mechanistic insight into how cytoskeleton organization is regulated during entosis remains poorly understood. The most consistent observation is that inhibition of Rho-ROCK signaling blocks entosis, indicating a central role for actomyosin contractility. Consistently, increased phosphorylation of myosin light chain and cortical actomyosin accumulation have been reported in invading cells, yet the underlying mechanisms remain largely unknown. Here, we systematically interrogated ROCK activation as a driver of the entotic internalization program. Constitutively active mutant ROCK1 or pharmacological ROCK1 activation using narciclasine rapidly triggered canonical entosis pathway through MLC2 phosphorylation. Mass spectrometry identified Plastin-3 (PLS3, T-plastin), a crucial calcium-sensitive actin-bundling protein, which was recruited to the plasma membrane to trigger entosis upon ROCK1 activation. Our study uncovers a key cytoskeletal remodeling step which may support cortical tension required for CIC invasion during entosis.