NBR1 shuttles between the cytoplasm and nucleus and is essential for nuclear p62 body formation
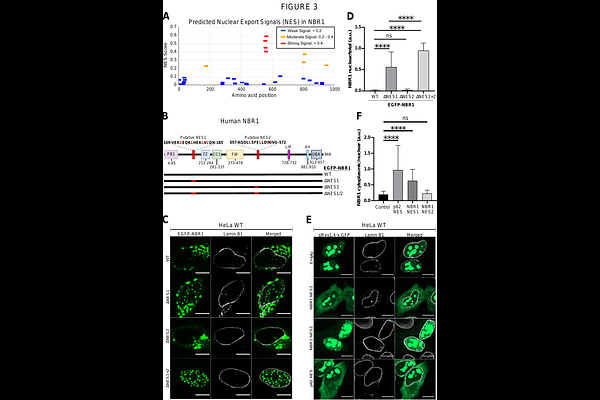
NBR1 shuttles between the cytoplasm and nucleus and is essential for nuclear p62 body formation
Moe Mauseth, M.; Wurz, J.; Svendsen Naess, M.; Evjen, G.; Olsvik, H. L.; Abudu, Y. P.; Johansen, T.; Lamark, T.
AbstractThe selective autophagy receptors SQSTM1/p62 and NBR1 are evolutionary related and involved in autophagy of different substrates including proteins, protein aggregates and organelles. The two proteins interact via their PB1 domains, and essential for their roles in autophagy is the formation of a specific type of condensates named p62 bodies. The scaffold of these structures is formed by the interaction of polymeric p62 with polyubiquitin, but NBR1 is recruited and essential for their formation. Previous studies have shown that p62 contains nuclear export signal (NES) and nuclear localization signal (NLS) motifs and shuttles between the cytoplasm and the nucleus. Its nuclear roles are not fully understood, but there is evidence that p62 is involved in protein quality control in the nucleus. No previous studies have tested if NBR1 is transported into the nucleus. We show here that NBR1 contains two NES motifs and one NLS motif, and like p62, the protein shuttles between the cytoplasm and the nucleus. NBR1 also accumulates in nuclear p62 bodies and the formation of nuclear p62 bodies depends on NBR1.