A codon-sensitive conformational switch gates commitment to translation start sites
A codon-sensitive conformational switch gates commitment to translation start sites
McGuire, S. F.; Chan, M. C.; Chan, T. C.; Pachikara, N.; Alleman, E. M.; Sikora, V. M.; Subramaniam, A. R.; Campbell, M. G.; Lapointe, C. P.
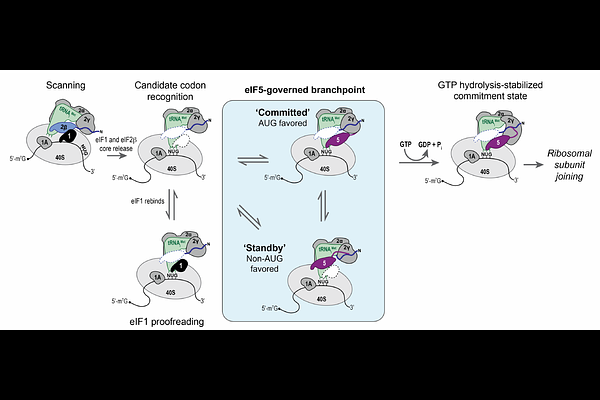
AbstractHuman translation initiation requires single-nucleotide precision to establish the reading frame, yet initiation at non-AUG codons plays key roles in gene expression. How the initiation machinery balances precision with this regulated flexibility remains unclear. Here, we define a conformational branchpoint governed by the human initiation factor eIF5 that gates commitment to start codons. Using single-molecule and structural approaches, we demonstrate that eIF5 reversibly occupies two conformations, which depends on a strictly conserved loop in the protein that monitors start codon identity. AUG codons favor the conformation that is stabilized by an eIF5-stimulated GTP hydrolysis step, which commits the complex to the start site. Non-AUG codons favor a standby conformation that destabilizes eIF5 and likely overlaps the binding site of an ancient structural homolog. This branchpoint complements enforcement of start codon fidelity by upstream steps and intrinsically controls the efficiency of non-AUG initiation.