A Translocation within the Ogataea Species Complex Alters Local Subtelomeric Chromatin while Maintaining Overall Genome Organization
A Translocation within the Ogataea Species Complex Alters Local Subtelomeric Chromatin while Maintaining Overall Genome Organization
Lundberg, T. J.; Lande, N. M.; Tourevski, D.; Figueroa, R.; Hanson, S. J.; Klocko, A. D.
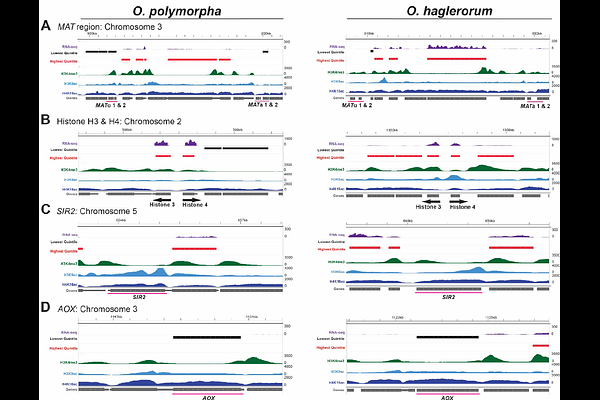
AbstractEukaryotic genomic DNA is packaged in the nucleus as chromatin - a DNA-protein aggregate regulating genome function, including transcription. Chromatin is classified as either active euchromatin or silent heterochromatin, with each marked by distinct histone post-translational modifications (PTMs). Chromatin composition also mediates genome organization, including how heterochromatin aggregates at the nuclear periphery while euchromatin localizes to the nucleus center. In fungi, heterochromatic loci cluster, including independent centromere and telomere clusters that form the Rabl chromosome conformation. However, it is unknown if chromatin composition and genome organization are conserved in closely related fungi, and how they are impacted by large-scale chromosomal rearrangements. Here, we examined differences in histone PTM deposition, gene expression, and genome organization in two yeast species from the order Pichiales, which diverged from the common ancestor shared with Saccharomyces cerevisiae more than 200 million years ago. We focused on Ogataea polymorpha, which is used for industrial protein production, and Ogataea haglerorum, an isolate of which harbors a translocation between chromosomes 1 and 6. We show that the enrichment of three activating PTMs - the trimethylation of lysine 4 of histone H3 (H3K4me3) and the acetylation of lysine 9 of histone H3 (H3K9ac) or lysine 16 of histone H4 (H4K16ac) - are similar genome-wide yet individual gene orthologs have distinct chromatin and expression patterns. While both Ogataea genomes organize into a Rabl conformation, the O. haglerorum translocation alters subtelomeric chromatin composition and expression of genes affected by the translocation. Our work highlights the genome function differences that occur on a microevolutionary scale.