ER-Lysosome Cholesterol Exchange Regulates Lysosomal Motility Through mTOR-Dependent LAMTOR1 Phosphorylation
ER-Lysosome Cholesterol Exchange Regulates Lysosomal Motility Through mTOR-Dependent LAMTOR1 Phosphorylation
Muthukottiappan, P.; Dehghani, A.; Muchamedin, A.; Ebner, M.; de Araujo, M. E. G.; Coman, C.; Rudnik, S.; Balachandran, M.; Fajardo-Callejon, S.; Akter, F.; Rösel, N.; Saftig, P.; Damme, M.; Ahrends, R.; Huber, L.; Haucke, V.; Gieselmann, V.; Winter, D.
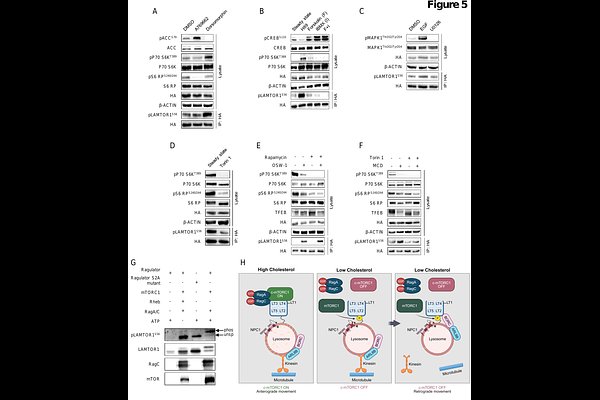
AbstractThe subcellular distribution of lysosomes, the main degradative organelles of mammalian cells, responds to metabolic cues in a highly dynamic way. While lysosomal positioning due to amino acid levels is well-characterized, cholesterol-dependent regulation of lysosomal motility is incompletely understood. We explored impaired lysosomal cholesterol export using a mass spectrometry-based multi-OMICs approach, identifying widespread reallocation of resources and signaling pathway modulation. We identified increased phosphorylation at LAMTOR1 serine 56 in response to cholesterol level perturbations. We demonstrate that this phosphorylation site is sufficient to disrupt Rag GTPases/SLC38A9 binding to the Ragulator complex, inhibiting canonical mTORC1 and facilitating binding of BORC, therefore promoting lysosomal retrograde movement. LAMTOR1 S56 phosphorylation responds exclusively to depletion of lysosomal limiting membrane cholesterol, is facilitated by mTOR, and presents a negative feedback loop for amino acid independent displacement of Ragulator bound Rag GTPases, limiting canonical mTORC1 activity. Mass spectrometry data are available via ProteomeXchange with identifier PXD073489.