Metabolostasis failure thresholds are linked with network topology, metabolite solubility, and translational control
Metabolostasis failure thresholds are linked with network topology, metabolite solubility, and translational control
A. Levkovich, S.; M Lim, C.; A. Marzini, E.; Adsi, H.; Lahav, M.; Sogolovsky-Bard, I.; Gartner, M.; Kaplan, K.; DeRowe, Y.; Pasmanik-Chor, M.; Brandis, A.; Vendruscolo, M.; Gazit, E.; Laor Bar-Yosef, D.
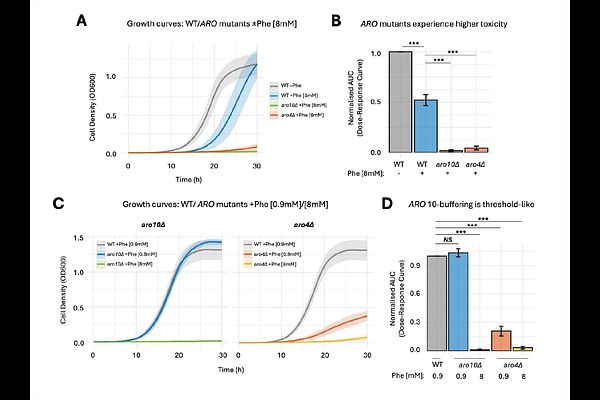
AbstractCells maintain metabolite homeostasis (metabolostasis) by buffering fluctuations in metabolite levels, yet the limits of this buffering and the mechanisms underlying metabolic toxicity remain poorly understood. To study this, we systematically overfed metabolites in Saccharomyces cerevisiae and quantified associations with growth inhibition, intracellular aggregation, and multiomic perturbations. We identify metabolite-specific failure thresholds at which amyloid-like aggregates are observed, with graded growth inhibition detectable at sub-threshold concentrations, suggesting toxicity mechanisms beyond transporter saturation. Metabolites with higher network influence and broader pathway participation are associated with higher failure thresholds and smaller pathway disturbances. These patterns are associated with chemical properties and solubility: more soluble metabolites, while broadly tolerated, are associated with localised aggregates at their failure thresholds, whereas less soluble metabolites are associated with larger systemic pathway disruptions. Multiomic integration identifies a two-tiered translational regulatory architecture characterising cellular resilience to metabolic overfeeding. General resilience is associated with transcriptional commitment to resource conservation via attenuation of anabolic pathways. Metabolite-specific defense is characterised by high-magnitude translational regulatory events; for example, engagement of aromatic catabolism under phenylalanine overfeeding and energetic control pathways under glycine overfeeding. Together, our results operationally define metabolostasis as a cellular system associated with constraint of metabolite concentrations, coordination of network and pathway-level regulation, and buffering against amyloid-like aggregation, highlighting how network topology, pathway architecture, and chemical properties are associated with metabolic resilience and toxicity thresholds.