De novo design of therapeutic scFvs and multi-specific engagers from sequence alone
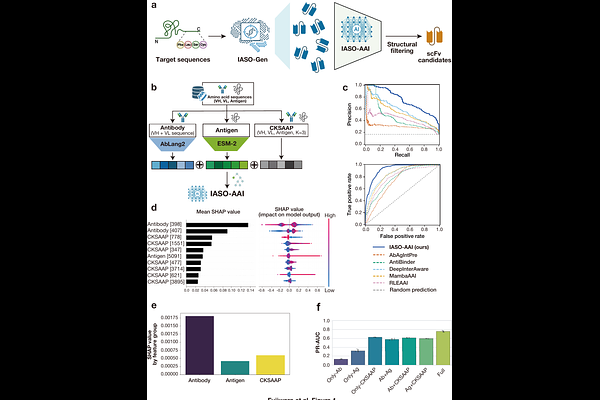
De novo design of therapeutic scFvs and multi-specific engagers from sequence alone
Fujiwara, T.; Shimizu, H.
AbstractThe discovery of functional antibody therapeutics is fundamentally constrained by the astronomical complexity of the sequence landscape and the inefficiency of empirical screening. While structure-based computational design has made significant strides, its reliance on high-resolution co-complex data restricts its utility against the vast undruggable proteome and rapidly evolving pathogens. Here, we introduce IASO, a sequence-centric generative framework for the de novo design of diverse biotherapeutic modalities, including single-chain variable fragments and multi-specific engagers, requiring only antigen sequence as input. IASO integrates an evolutionary-informed generative engine that constructs antigen-steered candidate libraries with a high-fidelity interaction module capturing synergistic physicochemical motifs. We demonstrate the framework's robustness by identifying novel binders against diverse clinical targets, where IASO successfully discriminates atomic-level mutations to overcome established drug resistance in EGFR and distinguishes single-residue variants of SARS-CoV-2. Furthermore, we extend this approach to the in silico construction of a complex bispecific T-cell engager that exhibits developability profiles comparable to clinically approved therapeutics, including enhanced solubility and minimal immunogenicity. By transforming serendipitous exploration into predictable high-resolution engineering, IASO establishes a scalable foundation for the rapid development of next-generation biopharmaceuticals directly from sequence data alone.