Structural basis of receptor retro-translocation in peroxisomal protein import
Structural basis of receptor retro-translocation in peroxisomal protein import
Dempsey, N. W. M.; Wang, L.; Gao, N.; Zhao, K.; Cope, J.; Park, E.
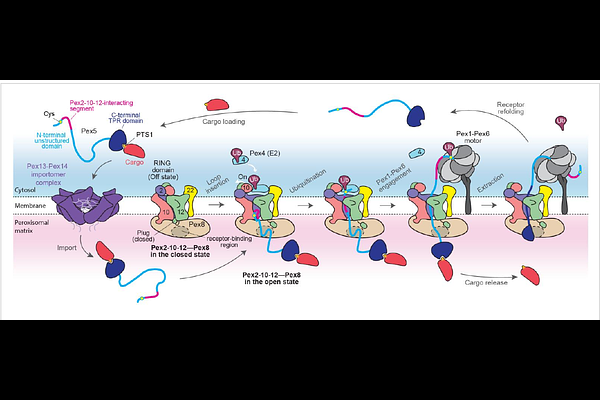
AbstractPeroxisomes import all matrix proteins post-translationally from the cytosol, a process that requires recycling of cargo receptors across the peroxisomal membrane. The membrane-embedded ubiquitin ligase, composed of Pex2, Pex10, and Pex12, is central to this process, but its mechanism remains unclear. Here we determined cryo-electron microscopy structures of the Saccharomyces cerevisiae Pex2-10-12 complex in closed and open states bound to Pex8, an essential factor of previously undefined function. The structures reveal how Pex2-10-12 gates its retro-translocation pore to control receptor entry and how the closed-to-open transition repositions the Pex10 RING domain to enable receptor mono-ubiquitination. Pex8 docks onto Pex2-10-12 from the matrix and guides receptors into the pore. Functional analyses show that the receptor's N-terminal segment downstream of its mono-ubiquitination site initiates a loop insertion into the pore. These findings establish how Pex2-10-12 coordinates receptor recognition, retro-translocation, and ubiquitination, providing the molecular basis for receptor recycling in peroxisomal protein import.