Interplay between Local Diffusion, Concentration, and Inter-Protein Alignment Promotes Cross-β-Sheet Transitions at Condensate Interfaces
Interplay between Local Diffusion, Concentration, and Inter-Protein Alignment Promotes Cross-β-Sheet Transitions at Condensate Interfaces
Castro, A.; Luengo-Marquez, J.; Tejedor, A. R.; Collepardo-Guevara, R.; Papp, M.; Arosio, P.; Ocana, A.; Sanchez-Burgos, I.; Rene Espinosa, J.
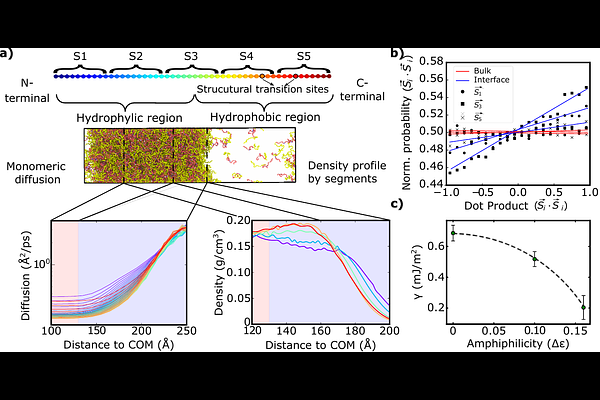
AbstractBiomolecular condensates govern cellular organization through dynamic, membraneless compartments whose material properties are dictated by intermolecular interactions. While liquid-liquid phase separation enables reversible condensate self-assembly, its dysregulation can trigger the formation of pathological solid-like states, often associated with neurodegenerative disorders. Here, we investigate the molecular mechanisms underlying condensate solidification, focusing on the role of inter-protein {beta}-sheet transitions. Using a minimal protein coarse-grained model, we reveal that the condensate's interface acts as a hotspot for inter-protein {beta}-sheet nucleation and growth. Residue- and segment-resolved analyses show that enhanced interfacial mobility, particularly of terminal domains, coupled with locally high protein concentrations, creates optimal conditions for inter-protein {beta}-sheet-sheet formation. Moreover, favourable inter-protein orientational alignment of terminal domains emerges at the interface. We find that asymmetric sequences-e.g. those with defined hydrophobic vs. more hydrophilic regions-further amplify this effect, resulting in local density fluctuations which concentrate aggregation-prone domains at the interface, and collectively promote inter-protein {beta}-sheet transitions. Such amphiphilic interfacial organization is driven by surface-tension minimization, and stabilizes protein structural rearrangements accentuating the spatial bias of inter-protein structural transitions. Altogether, our results demonstrate that general protein polymer-physics models, capturing only essential features such as flexibility and sequence patterning, inherently display condensate solidification at the interface. These findings identify the interface as a central regulator of condensate hardening, linking molecular-scale interactions to mesoscale phase behaviour, and providing mechanistic insight into the spatiotemporal onset of protein dysregulation in membraneless organelles.