Utilization of Cell-penetrating Peptide Adaptors to Enhance Delivery of Variably Charged Protein Cargos
Utilization of Cell-penetrating Peptide Adaptors to Enhance Delivery of Variably Charged Protein Cargos
Morris, D. P.; Turner, N. I.; Croffie, J. J.; McMurry, J. L.
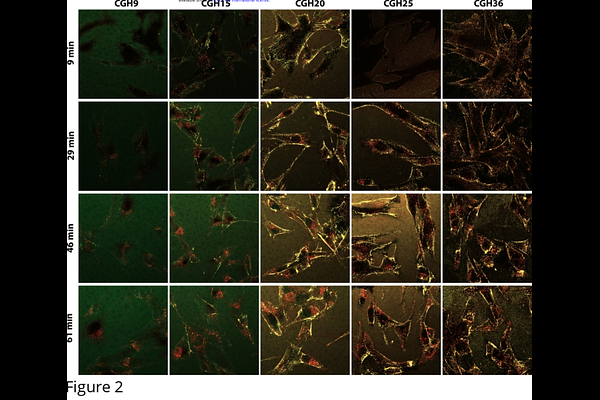
AbstractCell-penetrating peptides (CPPs) can deliver biomacromolecular cargos into cells, potentially enabling a new mode of intracellular drug delivery. However, a major problem with CPP-mediated delivery is entrapment of CPPs within endosomes and covalent linkages ensure CPPs and cargos share a common fate. We previously developed a CPP-adaptor system based on reversible, calcium-dependent cargo binding that produces cargo release from adaptors as complexes dissociate following internalization and Ca2+ efflux from early endosomes. Having employed CPP-adaptors with an array of protein cargos of differing charges, it became apparent that positively charged cargos often appeared to dominate internalization and that association with the adaptor had little effect. To systematically address the effects of cargo charge and CPP function, we tested the ability of several adaptors to increase internalization of a set of adaptor binding GFP cargos having net charges of +9, +15, +20, +25 and +36. Intrinsic internalization of these cargos reproduced reported patterns showing that positive charge increases internalization. However, labeling these cargos with a chemical fluorophore revealed that GFP fluorescence grossly underestimated total internalization. Internalization was charge and concentration dependent with more positive cargos showing apparent saturation of internalization at 100-400 nM, well below the concentrations at which covalently linked CPP-cargos are dosed. We tested the ability of 5 adaptors to internalize these cargos. Our prototype adaptor, TAT-CaM, was completely ineffective with the +9 cargo, but internalized moderately charged cargos extremely efficiently at concentrations far below the {micro}M range. A derivative adaptor, TAT-LAH4-CaM, was highly effective with all cargos and produced similar maximal internalization at 100-400 nM. However, two adaptors specifically designed with increased positive charge inhibited internalization of the most positive cargos. One of these, GFP-CaM, based on the supercharged GFP with net charge of +36, did increase internalization of the least positive cargos, demonstrating an adaptor with high affinity for the cell surface can increase internalization of a neutral cargo at very low concentration. The common maximal level of intrinsic GFP cargo internalization correlated with surface loading of these cargos, suggesting a limit to the beneficial effects of increased plasma membrane association. However, TAT-CaM further increased internalization via an apparently distinct mechanism. In this limited study of the interaction of cargo charge and adaptor efficacy, we found diverse behaviors that hint at the power and flexibility possible with adaptor/cargo internalization.