TRAILBLAZER: generative multicellular perturbation model of biology
TRAILBLAZER: generative multicellular perturbation model of biology
Grzybowski, A. T.; Nener, J.; Selvamani, P.; Badarinarayan, S. S.; Chandramohan, N.
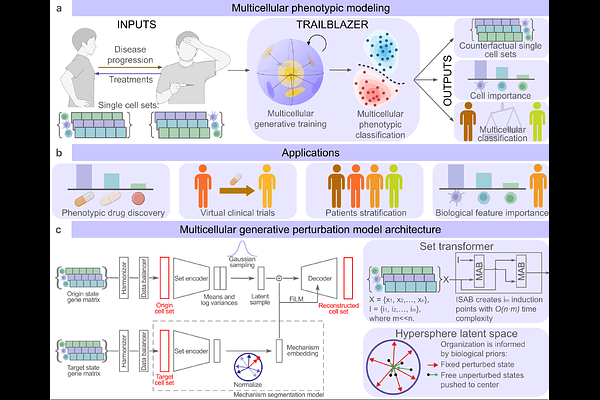
AbstractSingle-cell foundation models are reshaping biology by learning transferable representations of cellular state from millions of profiles. These models support annotation, denoising, cross-modal mapping and, increasingly, prediction of responses to genetic or pharmacological perturbations. Despite this progress, most approaches treat cells as independent observations and ignore the multicellular context that governs tissue behavior. Models trained on aggregated datasets often fail to generalize to new donors, laboratories or interventions, in part because their latent spaces lack structure for composition and extrapolation. As a result, strong reconstruction performance does not guarantee accurate forecasting of system-level responses. The general problem addressed here is how to construct a scalable model that predicts multicellular, patient-level responses to interventions while preserving single-cell resolution and enabling generalization beyond observed conditions. Here we show that TRAILBLAZER, a multicellular transformer encoder coupled to an explicitly shaped hyperspherical latent space and a count-aware generative decoder, enables accurate zero-shot prediction of perturbation responses and ranking of candidate immunomodulators at patient resolution. In contrast to prior single-cell or pseudo-bulk approaches, TRAILBLAZER models tissues as coordinated systems using latent tokens that summarize and redistribute global context while maintaining near-linear scaling with group size. By organizing latent geometry around shared healthy references and calibrated mechanistic directions, the model renders vector arithmetic biologically meaningful and supports extrapolation to unseen agents. Together, these results establish a practical framework for mechanism-aware simulation of multicellular responses and suggest a path toward predictive foundation models for therapeutic discovery.