Programmed electrical stimulation in human iPSC-derived cardiomyocytes reveals mechanisms of lethal arrhythmias in Calcium Release Deficiency Syndrome
Programmed electrical stimulation in human iPSC-derived cardiomyocytes reveals mechanisms of lethal arrhythmias in Calcium Release Deficiency Syndrome
Dababneh, S.; Arslanova, A.; Butt, M.; Halvorson, T.; Roston, T.; Roberts, J.; Ohno, S.; Jayousi, F.; Lange, P. F.; Hove-Madsen, L.; Rose, R. A.; Moore, E. D.; van Petegem, F.; Sanatani, S.; Chen, W. S. R.; Tibbits, G. F.; Prondzynski, M.
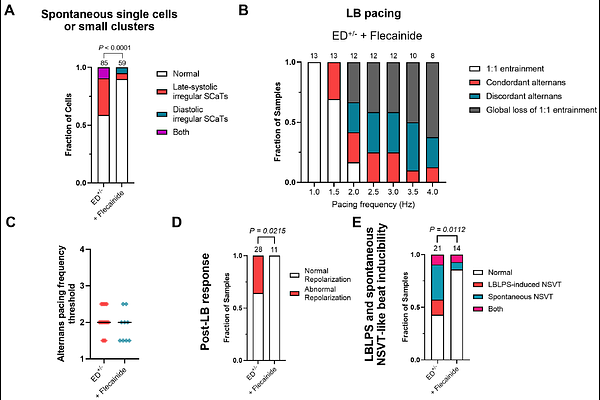
AbstractBackground: Calcium release deficiency syndrome (CRDS) is a recently described inherited channelopathy caused by loss-of-function variants in RYR2. Clinically, CRDS patients present with lethal ventricular arrhythmias which are not reproduced on exercise stress testing, unlike catecholaminergic polymorphic ventricular tachycardia. A hallmark trigger identified for CRDS mimics a long-burst, long-pause, short-coupled extra-stimulus (LBLPS) programmed electrical stimulation protocol, which was experimentally validated in humans and mouse models. Moreover, application of a long-burst, long-pause (LBLP) protocol alone can induce an abnormal repolarization on the first sinus beat that is unique to CRDS. However, the electrophysiological basis of CRDS in human cardiac tissue, including other triggers, are not fully understood, and whether clinically relevant arrhythmias can be observed in human stem cell models remains unknown. Methods: We performed electrophysiological and arrhythmia inducibility studies using clinically relevant programmed electrical stimulation protocols in two-dimensional cardiac tissue generated from metabolically matured human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) carrying the CRDS variant RyR2-E4146D. High spatiotemporal optical mapping and multielectrode arrays were used for electrophysiological phenotyping. Results: At baseline, E4146D+/- monolayers showed no arrhythmias, similar to controls. During rapid pacing, E4146D+/- promoted electrical vulnerability by reducing the threshold for action potential duration (APD) alternans and Ca2+ alternans and increasing the propensity for spatial discordance of alternans. In response to LBLP pacing, E4146D+/- monolayers often demonstrated an abnormal repolarization response characterized by spatially dispersed APD prolongation and large Ca2+ release. Notably, LBLPS pacing produced early-after depolarization (EAD)-driven triggered activity resulting in re-entrant tissue conduction patterns, explaining the short-coupled ectopy driven arrhythmias seen in CRDS patients. Similar arrhythmias were observed when EADs developed during spatially discordant alternans. Lastly, flecainide showed efficacy in suppressing arrhythmia inducibility for the here studied variant. Conclusions: We developed the first hiPSC model for CRDS which recapitulates clinically observed and inducible arrhythmias. Our model provides novel insights into tissue-level, re-entrant arrhythmias, which are initiated by EADs during electrically vulnerable states in CRDS human cardiac tissue and can be suppressed by flecainide. This model provides the framework for studying other CRDS variants and complex arrhythmias in hiPSC-CMs and establishes a human-based new approach method (NAM) for drug and gene therapy development for CRDS.