Enriched-GF: A Reproducible High-Yield Autologous Blood-Derived Growth Factor Method for Regenerative Medicine
Enriched-GF: A Reproducible High-Yield Autologous Blood-Derived Growth Factor Method for Regenerative Medicine
Bansal, H.; Singhal, M.; Bansal, A.; Khan, I.; Bansal, A.; Khan, S. H.; Leon, J.; al Maini, M.; Fernandez Vina, M.; Reyfman, L.
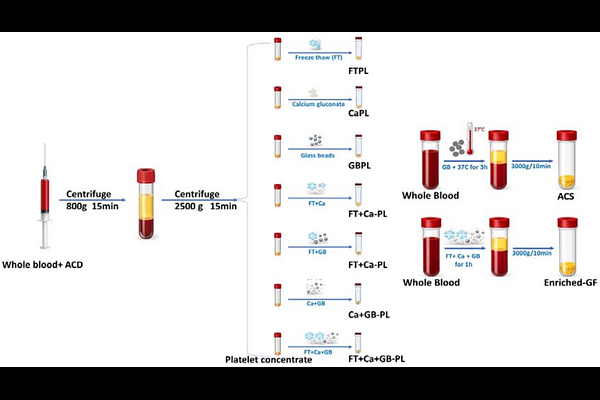
AbstractBackground Platelet-derived Growth factors play key roles in tissue repair and regeneration, yet conventional platelet-rich plasma (PRP) formulations release these mediators inconsistently in vivo due to variability in platelet yield and activation dynamics. To overcome this limitation, direct administration of concentrated platelet-derived growth factor preparations has gained interest, though current manufacturing approaches for human platelet lysate (hPL), growth factor concentrates (GFC), and conditioned serum remain constrained by batch variability, incomplete platelet degranulation, and reliance on anticoagulants. Here, we examine alternative platelet activation workflows to establish a standardized, efficient, and reproducible method for high-yield growth factor recovery suitable for translational and clinical applications. Methods Nine GFC production protocols were compared, employing different combinations of freeze thaw (FT) cycling, glass bead (GB) agitation, calcium (Ca) activation, and a novel Enriched Growth Factor (Enriched-GF) method. The objective was to identify a protocol capable of maximizing growth factor yield within a three-hour workflow. Optimal Ca concentrations and GB conditions were determined from prior optimization studies and integrated into the Enriched-GF processing scheme. Platelet concentrates (n = 10 per protocol) were processed under each condition, and growth factor levels were quantified using ELISA. Results Growth factor yields differed significantly across protocols. The greatest and most consistent increases in growth factor release were observed with the Enriched-GF method combining GB activation, FT cycling, and Ca stimulation. This approach resulted in markedly elevated concentrations of key regenerative mediators, including enhanced EGF release, a 4.5-fold increase in PDGF, maximal TGF{beta} liberation, and a four-fold increase in FGF2 relative to conventional platelet lysate or conditioned serum preparations. These results were reproducible across independent donor pools, demonstrating robustness and batch-to-batch consistency. Conclusion We describe a rapid and reproducible method for producing highly concentrated platelet-derived growth factors using a combined GB+FT+Ca activation strategy. The Enriched-GF protocol consistently outperformed existing platelet lysate, conditioned serum, and conventional GFC preparation methods, yielding a standardized product with enhanced growth factor content. This Enriched-GF approach offers a clinically practicable solution for applications in regenerative medicine requiring reliable and high-yield growth factor delivery. Keywords: Human Platelet Lysate; Growth Factor Concentrate; Regenerative Medicine; Platelet Activation.