Beyond Histology: A Unified Transcriptomic Atlas Defines Lung Cancer Biologic States and Subtypes
Beyond Histology: A Unified Transcriptomic Atlas Defines Lung Cancer Biologic States and Subtypes
Arora, S.; Suresh, L.; Thirmanne, H. N.; Jensen, M.; Glatzer, G.; Fatherree, J.; Konnick, E.; Levine, K.; Brooks, A. N.; Houghton, A. M.; Pritchard, C.; MacPherson, D.; Berger, A.; Holland, E. C.
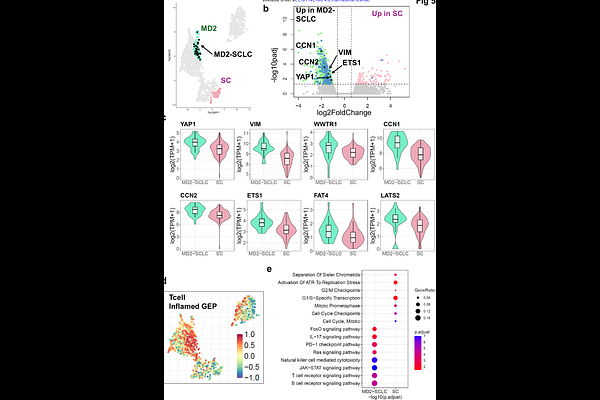
AbstractLung cancer encompasses multiple histological entities with substantial molecular heterogeneity that remain incompletely resolved at population scale. Here, we constructed a unified reference landscape of lung cancer by analyzing raw RNA sequencing data from 1,558 tumors spanning adenocarcinoma (n=753), squamous cell carcinoma (n=540), small cell lung cancer (n=150), and unclassified non-small cell lung cancer (n=80). Following batch correction, samples were embedded using PaCMAP to generate a continuous molecular atlas annotated with clinical and biological metadata. Rather than segregating strictly by histology, tumors organized along conserved transcriptional axes defined by tumor-intrinsic proliferative or metabolic programs and immune-infiltrated states. Consensus clustering resolved nine robust molecular clusters, including a female non-smoker-enriched adenocarcinoma subgroup, a neuroendocrine-like adenocarcinoma marked by ASCL1 activation, immune-associated regions, and bifurcation of both small cell and squamous carcinomas into biologically distinct states. Spatially-restricted expression of clinically actionable targets revealed state-specific vulnerabilities. Projection of patient tumors and patient-derived xenografts onto the atlas demonstrated preservation of transcriptional identity and enabled quantitative assessment of model fidelity. This unified framework redefines lung cancer as a structured continuum of transcriptional states with translational relevance.