CDK4/6 inhibitors enhance oxaliplatin efficacy in colorectal cancer with RB-dependent and tumor-selective activity in intestinal model
CDK4/6 inhibitors enhance oxaliplatin efficacy in colorectal cancer with RB-dependent and tumor-selective activity in intestinal model
Souza, A. S. O.; Conceicao, J. S. M.; Ferraz, L. S.; Delou, J. M. A.; Miranda, B. R.; Verissimo, C.; Carneiro, M. S. C.; Rehen, S.; Bonamino, M. H.; Borges, H. L.
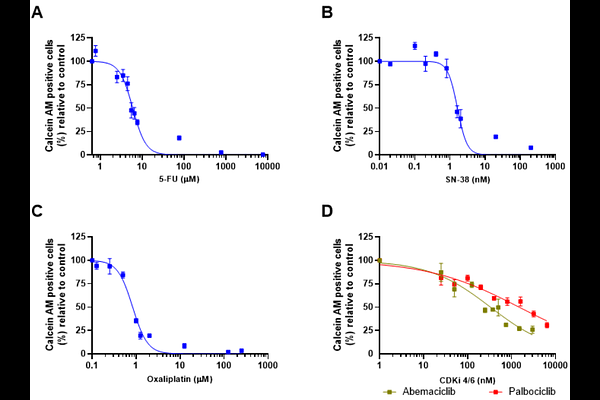
AbstractAlthough the retinoblastoma protein (pRB) is functionally inactivated by hyperphosphorylation in the majority of colorectal cancers (CRC) - with RB1 rarely mutated and even amplified at the genomic level - three critical gaps remain unaddressed: no study has systematically compared which first-line chemotherapeutic agent best synergizes with CDK4/6 inhibition using head-to-head quantitative analysis; functional differences between palbociclib and abemaciclib in chemotherapy combinations have not been characterized in CRC; and direct genetic evidence of RB dependency in this combinatorial context is lacking. Here, we addressed these gaps by evaluating palbociclib and abemaciclib combined with oxaliplatin, 5-fluorouracil, and SN-38 in HCT116 CRC cells, with validation in SW480 cells, RB1-silenced HCT116 cells (shRNA-RB), and non-tumoral intestinal epithelial cells (IEC-6), using quantitative drug interaction analysis (Chou-Talalay), cell cycle profiling, apoptosis assessment, and pRB phosphorylation measurement. Oxaliplatin was the most consistently synergistic partner for both CDK4/6 inhibitors (CI < 1 across all tested concentrations), while combinations with SN-38 yielded variable results and 5-FU combinations approached additivity. The oxaliplatin combination reinforced G1 arrest and enhanced cell death, with abemaciclib producing more pronounced apoptotic induction than palbociclib - an effect not explained by differential pRB target engagement (both inhibitors reduced pRB Ser807/811 phosphorylation by ~50%), likely reflecting abemaciclib's broader kinase inhibitory profile. shRNA-mediated RB1 silencing partially attenuated the combinatorial effect, providing direct genetic evidence that the synergy is RB-dependent. Importantly, the combination did not significantly potentiate oxaliplatin cytotoxicity in non-tumoral IEC-6 intestinal epithelial cells, in contrast to the pronounced enhancement observed in tumor cells, and synergistic benefit was preserved at sub-cytotoxic inhibitor concentrations. These findings identify oxaliplatin as the optimal chemotherapeutic partner for CDK4/6 inhibition in CRC, with a mechanism involving RB-dependent potentiation of apoptosis that is preferentially active against tumor cells and maintained at clinically relevant inhibitor doses.