Transitory enhancement of GATA2 chromatin engagement during early erythroid differentiation
Transitory enhancement of GATA2 chromatin engagement during early erythroid differentiation
Hobbs, J. W.; Taylor, S. J.; Kumari, R.; Haque, N.; Victor, L.; Steidl, U.; Coleman, R. A.
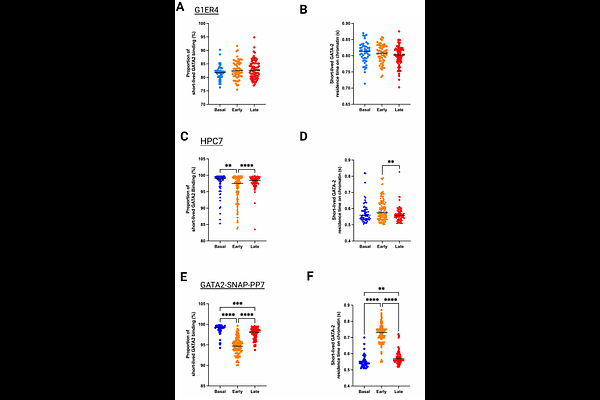
AbstractErythroid differentiation requires precise regulation of transcription factor binding to chromatin targets as hematopoietic progenitors relinquish multipotency and activate lineage programs. GATA2 maintains progenitor identity and is thought to be progressively silenced as GATA1 levels rise. However, the precise changes in GATA2 chromatin binding kinetics during this transition remain undefined. Here, we combined live-cell single-molecule imaging in cell lines and primary mouse progenitors with CUT&Tag chromatin profiling to define GATA2 activity during erythropoiesis. Single-molecule tracking resolved two interaction modes: short-lived (<1s) searching interactions and long-lived (>5 s) binding. Surprisingly, early erythroid differentiation was characterized by a transitory strengthening of long-lived GATA2 chromatin engagement. This manifested as increased residence time of GATA2 bound to chromatin in G1E-ER4 cells and an expansion of the long-lived bound population in HPC7 cells and primary mouse progenitors. This transitory phase of enhanced engagement declined upon further differentiation. Genome-wide mapping identified regulatory elements selectively occupied by GATA2 during this early transition state, revealing promoter-proximal sites enriched for GATA/RUNX motifs and distal elements containing composite GATA/E-box signatures. Together, our imaging and chromatin profiling indicate that GATA2 chromatin engagement is kinetically remodeled at the onset of differentiation, with early recruitment targets partitioning into distinct promoter- and enhancer-associated subclasses. These results support a model in which transcription factor kinetics constitute a dynamic chromatin engagement layer that characterizes the GATA2-to-GATA1 transition.