TriGraphQA: a triple graph learning framework for model quality assessment of protein complexes
TriGraphQA: a triple graph learning framework for model quality assessment of protein complexes
Liang, L.; Zhao, K.
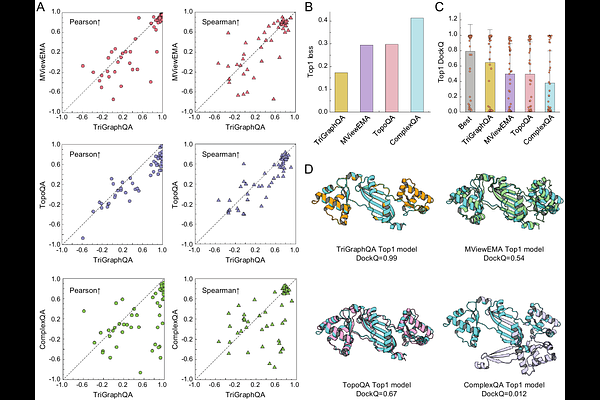
AbstractAccurate quality assessment of predicted protein-protein complex structures remains a major challenge. Existing graph-based quality assessment methods often treat the entire complex as a homogeneous graph, which obscures the physical distinction between intra-chain folding stability and inter-chain binding specificity. In this study, we introduce TriGraphQA, a novel triple graph learning framework designed for model quality assessment of protein complexes. TriGraphQA explicitly decouples monomeric and interfacial representations by constructing three geometric views: two residue-node graphs capturing the local folding environments of individual chains, and a dedicated contact-node graph representing the binding interface. Crucially, we propose an interface context aggregation module to project context-rich embeddings from the monomers onto the interface, effectively fusing multi-scale structural features. We conducted comprehensive tests on several challenging benchmark datasets, including Dimer50, DBM55-AF2, and HAF2. The results show that TriGraphQA significantly outperforms state-of-the-art single-model methods. TriGraphQA consistently achieves the highest global scoring correlations and lower top-ranking losses. Consequently, TriGraphQA provides a powerful evaluation tool for protein-protein docking, facilitating the reliable identification of near-native assemblies in large-scale structural modeling and molecular recognition studies.