NicheSphere reveals Spp1⁺ macrophages as central hubs coordinating fibrotic remodeling in myeloproliferative neoplasms
NicheSphere reveals Spp1⁺ macrophages as central hubs coordinating fibrotic remodeling in myeloproliferative neoplasms
Gleitz, H. F. E.; Ruiz Tejada Segura, M. L.; Nagai, J. S.; Fuchs, S. N. R.; Vroeg in de wei, G.; Snoeren, I.; Cesaro, G.; Bakker, I. J.; Gargallo Garasa, M.; Pritchard, J. E.; Klenovsek, T.; Schmitz, S.; Schmidt, L.; Bindels, E.; Lammers, T.; Gribnau, J.; Medyouf, H.; Schneider, M.; Schneider, C. V.; Kramann, R.; Gesteira Costa Filho, I.; Schneider, R. K.
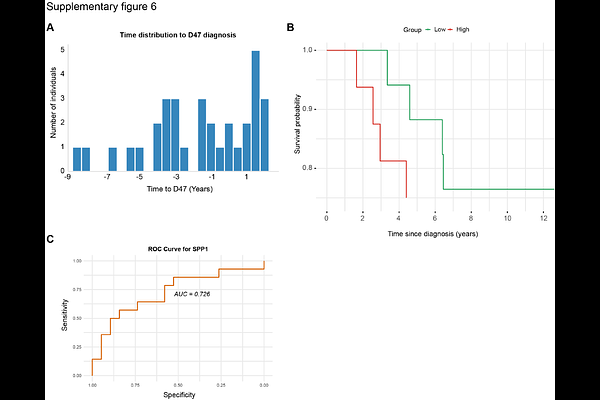
AbstractBone marrow fibrosis in myeloproliferative neoplasms arises from interactions between mutant hematopoietic clones and fibrosis-driving stromal cells. We identify Spp1 macrophages as central communication hubs integrating inflammatory and fibrotic programs via spatial proximity, ECM signaling, and cytokine activation. Using dual lineage-tracing, single-cell and multiplet RNA-sequencing, and a novel computational method for cell-colocalization and communication analysis named NicheSphere, we show that Spp1 macrophages form core communication hubs with osteoCAR cells, fibroblasts, and megakaryocytes. NicheSphere uncovered two distinct niches: macrophage-enriched compartments driving WNT, JAK-STAT, and TNF cytokine signaling, and a fibrosis-interacting core enriched in TGF-{beta} and ECM glycoproteins. Genetic ablation revealed cooperative roles of stromal and hematopoietic Spp1 in sustaining fibrosis and inflammation. Mechanistically, SPP1 promoted integrin-mediated adhesion, IL-1{beta} secretion, and stromal activation, while IL-1 cytokines induced Spp1 and collagen expression. Loss of Spp1 in hematopoietic progenitors reduced inflammation and restored macrophage function, establishing SPP1 macrophages as therapeutic targets in progressive bone marrow fibrosis.