Histidine exchange sustains LAT1 activity and proliferation in glutamine-addicted breast cancers.
Histidine exchange sustains LAT1 activity and proliferation in glutamine-addicted breast cancers.
Gjelaj, E.; Driscoll, P.; Mahmood, A.; Tarrago-Celada, J.; Kossifos, M.; Sesay, S.; MacRae, J. I.; Yuneva, M.
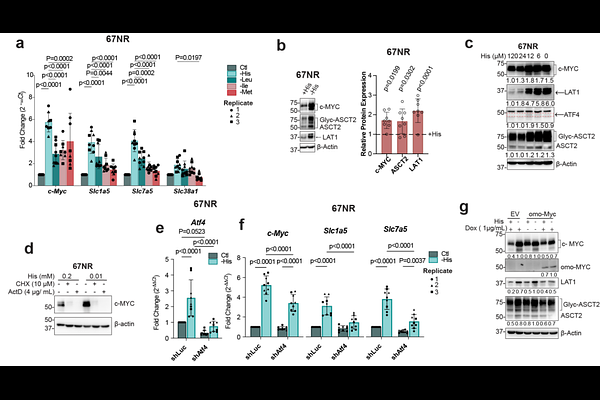
AbstractL-type amino acid transporter (LAT1) drives the uptake of essential amino acids (EAA) and activation of mTORC1 signaling in cancer cells. Current models propose that glutamine exchange is required for LAT1-dependent EAA transport; however, in many tumours, including MYC-driven cancers, glutamine is simultaneously consumed for bioenergetic and biosynthetic processes. How LAT1 activity is maintained under these conditions remains unclear. Here, we identify histidine as an efficient bidirectional LAT1 substrate that is preferentially utilised under glutamine-limitation in Gln-dependent tumour cells. Histidine uptake is modulated by glutamine availability, revealing an unexpected role for histidine in maintaining amino acid homeostasis. We demonstrate that histidine availability supports LAT1-mediated transport, promotes mTORC1/4E-BP1 signaling, and enhances protein synthesis and supports tumour cell proliferation. Under histidine limitation, MYC and ATF4 induce amino acid transporter expression, including LAT1, to preserve intracellular EAA levels. Importantly, histidine restriction sensitizes tumour cells to LAT1 inhibition, enhancing sensitivity to LAT1 inhibition and reducing tumour burden. Together, our findings establish histidine as a key regulator of LAT1 function and mTORC1 activity, suggesting a potential metabolic vulnerability in glutamine-dependent tumours.