Beyond thermal unfolding: urea-gradient nanoDSF approach for thermostability analysis of kinetically stable hyperthermophilic proteins
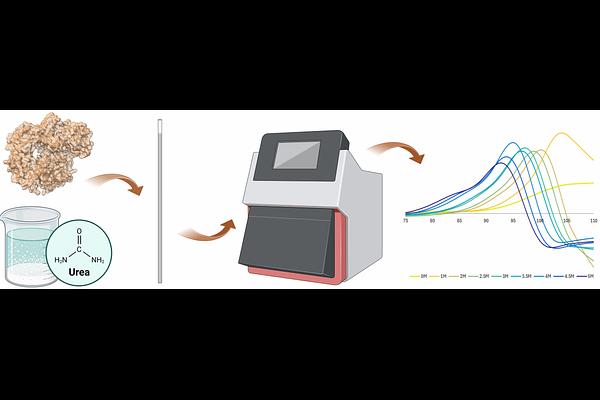
Beyond thermal unfolding: urea-gradient nanoDSF approach for thermostability analysis of kinetically stable hyperthermophilic proteins
Rusinek, W.; Dorawa, S.
AbstractIn this study, we demonstrate that urea enables reliable melting temperature (Tm) determination of hyperthermostable proteins by nano differential scanning fluorimetry (nanoDSF) Under native conditions, Pfu DNA polymerase and its Sso7d-fusion variant showed no detectable unfolding transitions, despite their Tm values falling within the instruments operational range, reflecting their extreme kinetic stability. In the presence of up to 7 M urea, intrinsic tyrosine and tryptophan fluorescence revealed clear unfolding transitions, yielding extrapolated Tm values of 104.8 {+/-} 0.09 {degrees}C for Pfu and 106.8 {+/-} 0.33 {degrees}C for its Sso7d-fusion variant. These results demonstrate that urea-gradient nanoDSF overcomes both instrumental and kinetic limitations, providing a simple and robust method for assessing the thermal stability of (hyper)thermostable proteins.