A Divergent Class of Arylamine N-Acetyltransferases Catalyzes Convergent Amidative Condensation of Polyketides in Manumycins Biosynthesis
A Divergent Class of Arylamine N-Acetyltransferases Catalyzes Convergent Amidative Condensation of Polyketides in Manumycins Biosynthesis
Yan, X.; Yan, G.; Ma, B.; Zhou, Q.; Luo, M.; Wei, G.; Lin, Z.; Deng, Z.; Kong, X.; Qu, X.
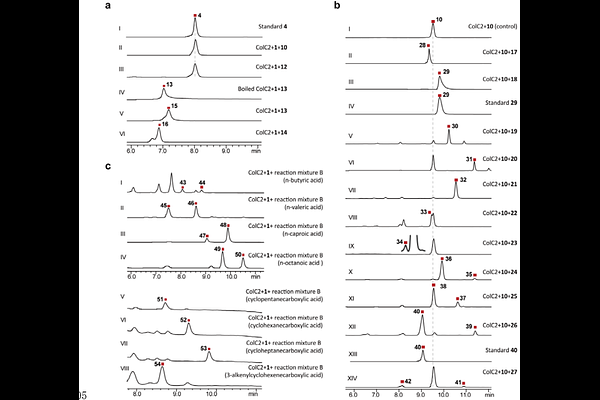
AbstractPolyketides are prized for their structural complexity and therapeutic potential, yet the incorporation of amide bonds into their frameworks typically relies on linear, nonribosomal peptide synthetase (NRPS)-dependent assembly. The direct, convergent coupling of distinct polyketide chains via amide bond formation -- an ideal strategy for combinatorial biosynthesis -- has remained largely elusive. Here, we report the discovery of a novel family of arylamine N-acetyltransferases (NATs) from manumycin-type biosynthetic pathways that catalyze an unprecedented intermolecular amidative chain transfer/condensation between an acyl carrier protein (ACP)-tethered polyketide donor and a free polyketide acceptor. Biochemical, structural, and molecular dynamics studies reveal that the representative enzyme, ColC2, possesses a distinctive substrate-binding pocket that diverges from canonical arylamine NATs, conferring exceptional promiscuity toward diverse acyl donors and acceptors. We demonstrate the utility of this biocatalyst by coupling arylamines with either synthetic acyl-thioesters or polyketide synthase (PKS) machinery to generate a library of non-natural polyketide amides and manumycin derivatives. These findings establish a new paradigm for amide bond formation in polyketide biosynthesis and position arylamine NATs as powerful tools for the development of novel therapeutics through combinatorial synthesis.