Structural studies of KCTD1 and its disease-causing mutant P20S provide insights into the protein function and misfunction
Structural studies of KCTD1 and its disease-causing mutant P20S provide insights into the protein function and misfunction
Balasco, N.; Ruggiero, A.; Smaldone, G.; Pecoraro, G.; Coppola, L.; Pirone, L.; Pedone, E. M.; Esposito, L.; Berisio, R.; Vitagliano, L.
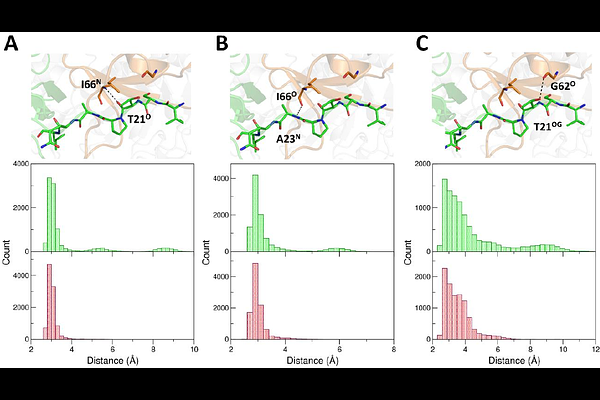
AbstractMembers of the KCTD protein family play key roles in fundamental physio-pathological processes. A plethora of literature studies have demonstrated their involvement in cancer, neurodevelopmental disorders, and genetic diseases. Despite two decades of intense investigations, the definition of structure-(mis)function relationships for these proteins is still rather limited. Here, we derived atomic-level structural data on KCTD1, by determining the crystal structure of its P20S mutant, which causes the scalp-ear-nipple syndrome, and performing molecular dynamics simulations. In addition to the expected folded domains (BTB and CTD) the crystal structure unravels that also the N-terminal region that precedes the BTB domain (preBTB) adopts a folded polyproline II (PPII) state. The global structure of the KCTD1 pentamer is characterized by an intricate architecture in which the different subunits mutually exchange domains to generate a closed domain swapping motif. In this framework, the BTB domain of each chain makes peculiar contact with the preBTB and the CTD regions of an adjacent chain. Indeed, the BTB-preBTB interaction is made of a PPII-PPII recognition motif whereas the BTB-CTD contacts are mediated by an unusual (+/-) helix discontinuous association. The inspection of the protein structure, along with the data that emerged from the MD data, provides a clear explanation of the pathogenicity of the SENS mutation P20S and unravels the role of the BTB-preBTB interaction in the insurgence of the disease. Finally, the presence of potassium bound to the central cavity of the CTD pentameric assembly provides insights into the role of the protein in metal homeostasis.