A Global Ligandability Map of Tryptoline Butynamide Stereoprobes Identifies Covalent Inhibitors of the Actin Maturation Protease ACTMAP
A Global Ligandability Map of Tryptoline Butynamide Stereoprobes Identifies Covalent Inhibitors of the Actin Maturation Protease ACTMAP
Xiong, Y.; Reinhardt, C. J.; Nguyen, T.; Hoffman, M. A.; Simon, G. M.; Melillo, B.; Cravatt, B. F.
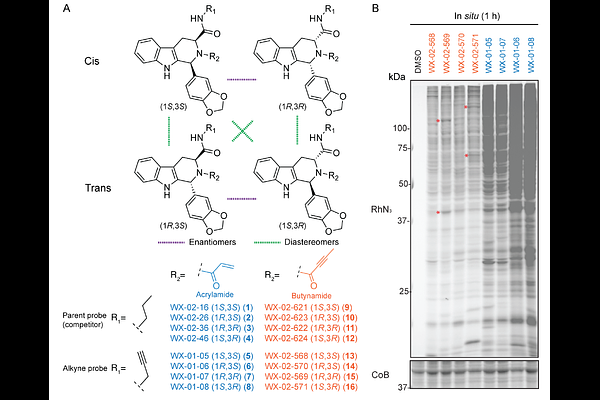
AbstractCovalent chemistry coupled with activity-based protein profiling (ABPP) offers a versatile approach for small-molecule ligand discovery in native biological contexts. The covalent ligandability maps generated by ABPP that target cysteine have frequently leveraged the acrylamide as a reactive group due to its tempered electrophilicity and presence in many advanced tool compounds and therapeutics. More recently, alternative cysteine-directed re-active groups such as the butynamide have emerged as an additional source of covalent probes and drugs, but their global reactivity with the proteome remains largely unexplored. Here, we compare the ligandability maps of stereochemically defined acrylamide and bu-tynamide compounds (stereoprobes) built from a common tryptoline core and find that the butynamides, despite exhibiting attenuated intrinsic and proteome-wide reactivity, prefer-entially engage a diverse set of proteins in human cancer cells. Among the butynamide-preferring proteins was C19orf54/ACTMAP, a cysteine protease required for the post-translational maturation of actin. We show that (1S, 3R)-tryptoline butynamides stereose-lectively react with the catalytic nucleophile of ACTMAP, leading to accumulation of N-terminally unprocessed actin in cancer cells. Our findings support reactive group diversifi-cation as a strategy for expanding the ligandability of the human proteome and the bu-tynamide, more specifically, as a differentiated cysteine-directed electrophile for chemical probe discovery.