Structural Basis of M1 Muscarinic and H3 Histamine Receptor Inhibition in OPC Differentiation
Structural Basis of M1 Muscarinic and H3 Histamine Receptor Inhibition in OPC Differentiation
Raubenolt, B.; Cumbo, F.; Joshi, J.; Martin, W.; Medicetty, S.; Yang, Y.; Trapp, B.; Blankenberg, D.
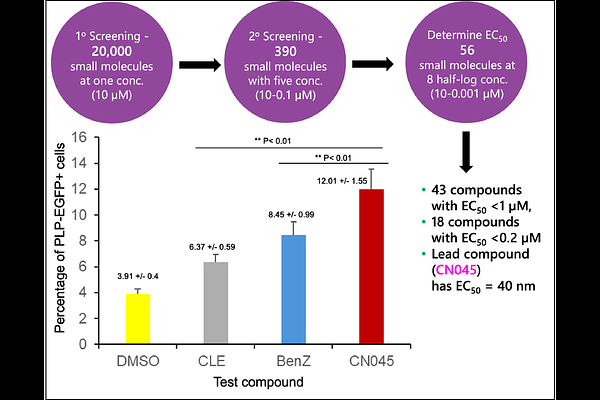
AbstractMuscarinic and histamine receptors are neurotransmitter-binding proteins within the large family of G protein-coupled receptors (GPCRs) and are relevant to human health and disease, including multiple sclerosis (MS), a chronic immune-mediated inflammatory demyelinating disease of the central nervous system (CNS) with neurodegenerative components. MS affects approximately 1 in 333 people, and women are affected at roughly threefold higher rates than men. A major pathological feature of MS is demyelination with incomplete remyelination of axons in the CNS. Because oligodendrocyte progenitor cells (OPCs) can differentiate into mature oligodendrocytes that restore myelin, small molecules that promote OPC differentiation represent a potential therapeutic strategy. High-throughput screening identified 18 hit compounds with EC50 values below 0.2 M, including the lead compound CN045, which showed an EC50 of 40 nM in vitro. Cheminformatic and experimental target identification studies implicated the M1 muscarinic receptor and the H3 histamine receptor as candidate targets. To interpret these findings, we performed docking, molecular dynamics simulations, and binding free-energy analyses on complexes involving CN045 and clemastine, a known antihistamine with antimuscarinic activity. The simulations support weaker and less stable binding of CN045 to H3 than to M1 and identify residue-level interactions that contribute to stability within the M1 binding pocket. Comparisons between CN045 and clemastine at M1 further suggest that the two ligands sample different local conformational ensembles, including differences in conserved microswitch behavior associated with active-like versus inactive-like receptor states. Together, these results provide a structural framework for understanding ligand-specific M1 engagement and may help guide future optimization of remyelination-promoting compounds.