Domain-Specific Agonist Binding Affinities Explain Structural and Functional Regulation of TRPM2
Domain-Specific Agonist Binding Affinities Explain Structural and Functional Regulation of TRPM2
Kupriianova, T.; Schwarzer, T.; Thalacker, T.; Defelipe, L.; Etzold, S.; Kulow, F.; Pahl, V.; Goyal, S.; Nguyen, V.; Zimmermann, M.; Guse, A.; Cambronne, X. A.; Tidow, H.; Fliegert, R.; Garcia-Alai, M. M.
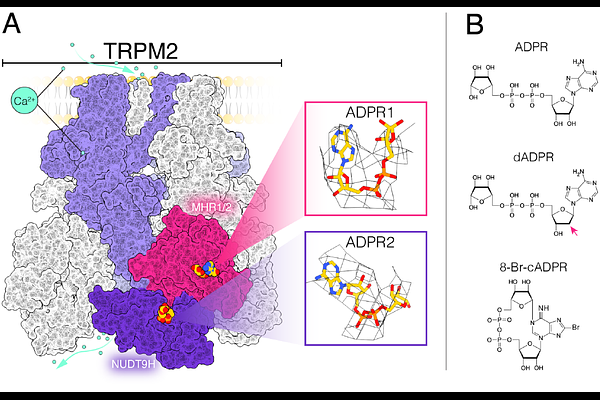
AbstractTRPM2 is a Calcium permeable cation channel activated by ADP-ribose (ADPR) and oxidative stress, yet the relative contributions of its two nucleotide-binding domains, MHR1/2 and NUDT9H, remain incompletely understood. Here, we quantitatively determine the affinities of the isolated human TRPM2 MHR1/2 and NUDT9H domains for ADPR, 2-deoxy-dADPR (dADPR), and 8-Br-cADPR using biophysical approaches. The MHR1/2 domain binds ADPR with high affinity (Kd; 0.5 uM), whereas the NUDT9H domain displays substantially lower affinity (Kd; 192 uM), revealing a difference of nearly three orders of magnitude. Mutational analysis demonstrates that alterations in MHR1/2 strongly affect ligand binding and channel activation, while mutations within NUDT9H that markedly reduce ligand affinity exert only modest effects on gating. In parallel, we quantify intracellular ADPR concentrations in resting and hydrogen peroxide stimulated cells and find that they remain well below the affinity required for substantial NUDT9H occupancy. Together, our findings indicate that high-affinity binding to the MHR1/2 domain is sufficient to drive TRPM2 activation under physiological conditions, whereas the NUDT9H domain likely contributes to maintaining the structural integrity of the channel rather than directly mediating ligand-dependent activation. These results provide a quantitative framework for understanding ligand-dependent TRPM2 regulation in cells.