Tetrahydrocurcumin Suppresses Bladder Carcinogenesis via Reprogramming O-GlcNAcylation-Phosphorylation Crosstalk
Tetrahydrocurcumin Suppresses Bladder Carcinogenesis via Reprogramming O-GlcNAcylation-Phosphorylation Crosstalk
Yang, M.; Li, R.; Zhou, M.; Dong, Y.; Zhao, J.; Tan, R.
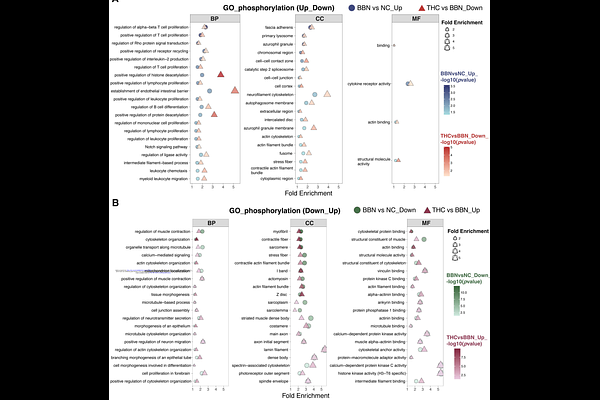
AbstractBladder cancer exhibits high recurrence rates and limited therapeutic options in advanced stages. Although post-translational modifications (PTMs) are critical regulators of tumor biology, their systems-level remodeling during bladder carcinogenesis remains insufficiently defined. Using a BBN-induced murine bladder cancer model combined with tumor-derived organoids and bladder cancer cell lines, we observed a consistent global elevation of protein O-GlcNAcylation across tumor-associated contexts. Site-resolved quantitative proteomics revealed that this increase reflected selective redistribution rather than uniform accumulation, with differential O-GlcNAc sites nearly evenly divided between up- and down-regulated events. Integrated analyses demonstrated preferential enrichment of up-regulated O-GlcNAcylation within cytoskeleton-adhesion modules and Notch-related domains, whereas down-regulated events were more closely associated with structural maintenance and homeostatic programs. Approximately half of differentially O-GlcNAcylated proteins exhibited concurrent phosphorylation changes, indicating coordinated multi-layer PTM remodeling. Pharmacological inhibition of O-GlcNAc transferase suppressed bladder cancer cell proliferation and organoid growth, supporting a functional association between elevated O-GlcNAcylation and tumor growth. Tetrahydrocurcumin (THC) reduced global O-GlcNAcylation and induced directional remodeling of BBN-associated O-GlcNAc patterns. Cross-comparative analyses identified subsets of sites exhibiting opposite regulation trends following THC intervention. Collectively, these findings define a spatially and functionally organized PTM remodeling landscape in bladder cancer and suggest that THC exerts anti-tumor effects in part through coordinated reprogramming of O-GlcNAcylation and phosphorylation networks.