BAF complexes maintain accessibility at stimulus-responsive chromatin and are required for transcriptional stimulus responses
BAF complexes maintain accessibility at stimulus-responsive chromatin and are required for transcriptional stimulus responses
Gulka, A. O. D.; Kang, K. A.; Zhou, Z.; Gorkin, D. U.
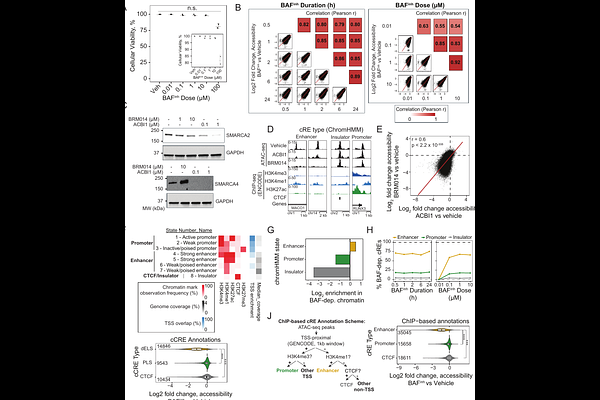
AbstractBackground Gene expression changes in response to developmental and environmental cues rely on cis-regulatory sequence elements (cREs). BRG1/BRM-Associated Factors (BAF) chromatin remodeling complexes maintain chromatin accessibility at many cREs, enabling binding by transcription factors (TFs). However, cREs exhibit a broad range of sensitivity to loss of BAF function, and the basis of this variability remains unknown. Results To identify the characteristics of BAF-dependent cREs, we mapped chromatin accessibility changes following acute pharmacologic BAF inhibition in GM12878 lymphoblastoid cells. We integrated these results with over 100 TF and histone modification ChIP-seq datasets and used machine learning to identify features that predict chromatin accessibility changes. We found that Activator Protein 1 (AP-1) factors and lymphoid lineage-defining TFs including RUNX3 and PU.1 predicted BAF-dependence. Strikingly, we found that cREs bearing the chromatin signature of "primed" enhancers - enriched for H3K4me1 but lacking H3K27ac - were significantly more sensitive to BAF inhibition than typical active enhancers. As primed enhancers are known to facilitate transcriptional responses to stimuli, we tested the requirement of BAF activity in these responses. Acute BAF inhibition was sufficient to prevent both chromatin and transcriptional responses to interferon gamma and dexamethasone. cREs which normally gained accessibility in response to stimulation failed to do so with BAF inhibition, and these cREs were linked to genes with suppressed transcriptional induction. Conclusions Collectively, our results demonstrate a requirement for continuous BAF activity to enable stimulus response and suggest that defective signal responsiveness may be a pathogenic mechanism in disease states caused by loss-of-function mutations in BAF subunits.