Proteomic mapping of novel tubulin post-translational modifications in Trypanosoma cruzi cytoskeleton
Proteomic mapping of novel tubulin post-translational modifications in Trypanosoma cruzi cytoskeleton
Martinez Peralta, G.; Baldelomar, D.; Baldasseroni, L.; SERRA, E.; Alonso, V. L.
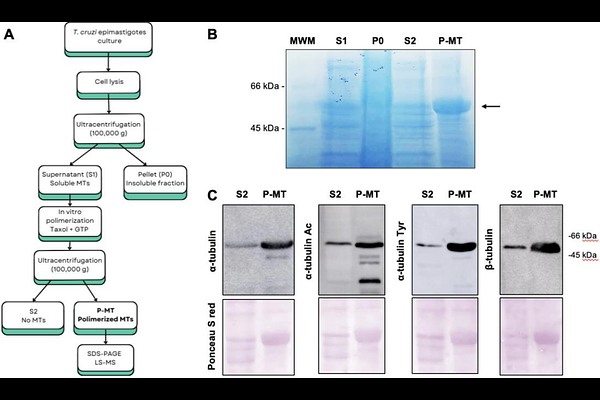
AbstractMicrotubules (MTs) play central roles in the organization and morphology of trypanosomatid parasites, forming highly specialized cytoskeletal structures such as the subpellicular corset, the flagellar axoneme, and the mitotic spindle. Functional specialization of MTs is regulated by the "tubulin code", which is defined by the combination of different - and {beta}-tubulin isotypes, a set of post-translational modifications (PTMs) and specific MT-binding proteins. Although multiple tubulin PTMs have been described in trypanosomatids using specific antibodies or mass spectrometry, to date no comprehensive mapping has been reported in Trypanosoma cruzi, the causative agent of Chagas Disease. In the present work, we performed a high-resolution proteomic analysis of PTMs present in - and {beta}-tubulin subunits of the T. cruzi Dm28c strain, using tubulin-enriched extracts obtained by in vitro polymerization. Multiple PTMs were identified, including acetylation, methylation, phosphorylation, and polyglutamylation, for which many modified amino acids had not been previously reported in trypanosomatids. Structural mapping of these modifications onto a predicted /{beta}-tubulin heterodimer showed that most modified residues are located in solvent-exposed regions of the protein. Together, these findings provide the first systematic map of tubulin PTMs in T. cruzi and support the existence of a complex tubulin code contributing to microtubule regulation in this parasite.