Stromal asparagine supports tumor adaptation to oxidative phosphorylation inhibition through SLC38A4-mediated metabolic coupling
Stromal asparagine supports tumor adaptation to oxidative phosphorylation inhibition through SLC38A4-mediated metabolic coupling
Qin, Z.; Li, S.; Xu, Y.; Zou, J.; Ma, J.; Wang, Y.; Wang, Y.; Ju, R.; Wang, L.; Guo, L.
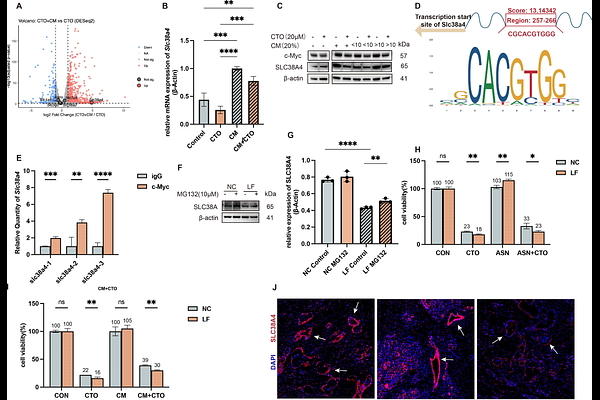
AbstractPurpose Pancreatic ductal adenocarcinoma (PDAC) is characterized by a nutrient-deprived and hypoxic tumor microenvironment (TME) that imposes severe metabolic stress on cancer cells. Under these conditions, tumor cells frequently activate the integrated stress response (ISR) to adapt to TME and develop resistance to therapies. However, how TME components support tumor adaptation to mitochondrial metabolic stress remains incompletely understood. Here, we aimed to identify key metabolite involved in ISR adaptation under oxidative phosphorylation (OXPHOS) inhibition and to elucidate the metabolic symbiosis between cancer-associated fibroblasts (CAFs) and PDAC cells. Methods We integrated transcriptomic and metabolomic analyses with functional assays. ISR activation was evaluated by assessing the phosphorylation of eIF2 (p-eIF2) following treatment with carboxyamidotriazole orotate (CTO), an Complex I inhibitor. Metabolomic profiling was used to identify metabolites involved in ISR activation alleviation. Mouse models were used to assess therapeutic responses following depletion of the identified metabolite under CTO treatment. Genetic perturbation of Slc38a4 was performed to assess its functional role in tumor cell adaptation to metabolic stress. Results We identified asparagine (ASN) as a critical metabolite supplied by CAFs to PDAC cells under OXPHOS inhibition. A minimum level of ASN is required for PDAC cells to execute ISR downstream adaptation. ASN depletion significantly enhanced the anti-tumor efficacy of OXPHOS inhibition both in vitro and in vivo. SLC38A4 emerged as a potential mediator of this interaction. SLC38A4 expression was associated with c-Myc, and its loss increased the sensitivity of PDAC cells to CTO-induced metabolic stress. Conclusion Our findings reveal a CAF-tumor metabolic crosstalk in which stromal-derived ASN supports PDAC cell adaptation to mitochondrial metabolic stress. Adaptive outcome of ISR signaling depends on the availability of key metabolic substrates such as ASN. When extracellular ASN supply is limited, the ATF4-dependent adaptive program collapses, converting ISR from a pro-survival response into a therapeutic vulnerability. SLC38A4 may function as a key mediator of this metabolic coupling and represents a potential target for enhancing the efficacy of OXPHOS inhibition in PDAC.