Synthetic circRNAs employ IRES activity for translation in cells and in cell-free translation systems
Synthetic circRNAs employ IRES activity for translation in cells and in cell-free translation systems
Koch, P.; Arendrup, F. S. W.; Lim, C.; Narayanan, S.; Adam, A.; Clamer, M.; Lund, A. H.; Chen, C.-K.; Leppek, K.
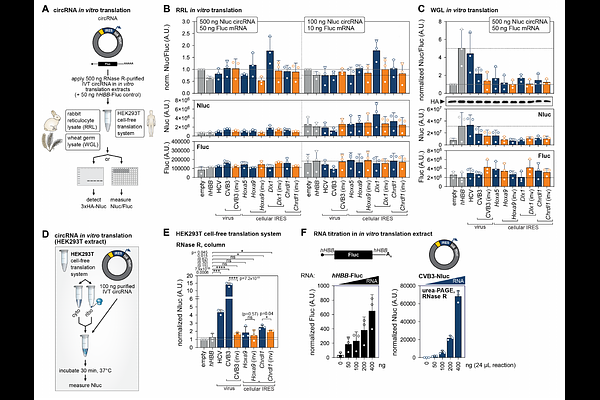
AbstractGene regulation through translation is critical for spatiotemporal protein expression. Internal ribosomal entry sites (IRESes) mediate mRNA-specific translation by recruiting ribosomes to 5' untranslated regions. Circular RNAs (circRNAs), naturally occurring and stable RNA species, are increasingly used as synthetic tools for sustained therapeutic protein translation by IRES-driven initiation. However, the functionality of different IRESes in synthetic circRNAs remains sparsely characterized. We systematically examine circRNA reporter translation by viral and cellular IRESes in human cells and in diverse in vitro translation systems. Improved circRNA purification by urea-PAGE and RNase R-treatment removes contaminants that induce RNA sensing. Viral CVB3 and HCV, as well as cellular Hoxa9, Chrdl1, Cofilin and c-Myc IRESes, effectively drive circRNA translation. We also establish circRNA translation in an improved human cell-free extract that recapitulates IRES-dependent regulation, and allows for precise engineering of HCV IRES-mediated translation. These findings inform IRES selection for synthetic circRNA translation relevant for circRNA-based medicines.