Mechanistic insights into the association and activation of the SARS-CoV-2 2'-O-Methyltransferase (NSP16)
Mechanistic insights into the association and activation of the SARS-CoV-2 2'-O-Methyltransferase (NSP16)
Ma, H.; Brace, A.; Lemus, M. R.; Chennubhotla, S. C.; Satchell, K. J.; Ramanathan, A.
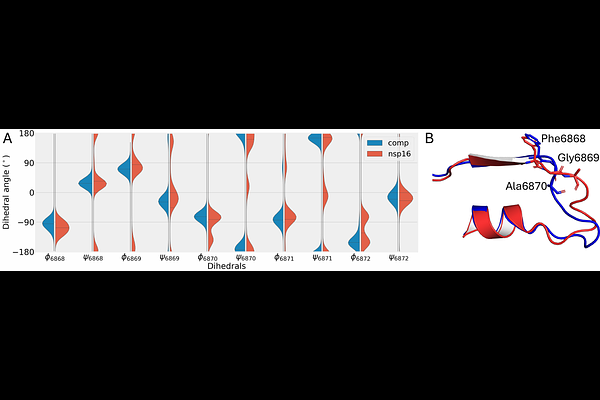
AbstractThe nsp16 2'-O-Methyltransferase is an essential non-structural protein of SARS-CoV-2, which methylates the viral mRNA cap structure, enabling it to evade the host immune response for higher translation efficiency. However, nsp16 is only active when it is bound to its cofactor, namely the non-structural protein-10 (nsp10). Understanding how nsp10 binds to and activates nsp16 function can help to develop targeted inhibitors; however, given the varying degree of disorder in both nsp10 and nsp16, characterizing this interaction has been challenging. Using long-timescale molecular dynamics simulations and AI/ML methods, we posit that the nsp16/nsp10 binding process is mediated by a hydrophobic latch formed with Leu4298 from nsp10 and a hydrophobic concave on the nsp16 protein surface. Our study highlights how the nsp16 S-adenosyl-L-methionine (SAM) pocket closes in its monomer state, which in turn deactivates the MTase function. We also observe that the nsp16/nsp10 complex allows for the RNA binding site to open with the empty SAM pocket. The results reveal how the SAM pocket loops facilitate SAM binding while allowing for the by-product S-adenosyl-L-homocysteine (SAH) to exit. Our study thus provides valuable atomistic-level mechanistic insights into understanding the activation of nsp16 MTase function while highlighting the challenges of studying protein-protein interactions mediated by largely flexible/disordered regions.