RGS6 regulates Kappa Opioid Receptor-mediated antinociceptivebehaviors
RGS6 regulates Kappa Opioid Receptor-mediated antinociceptivebehaviors
Blount, A.; Sutton, L.
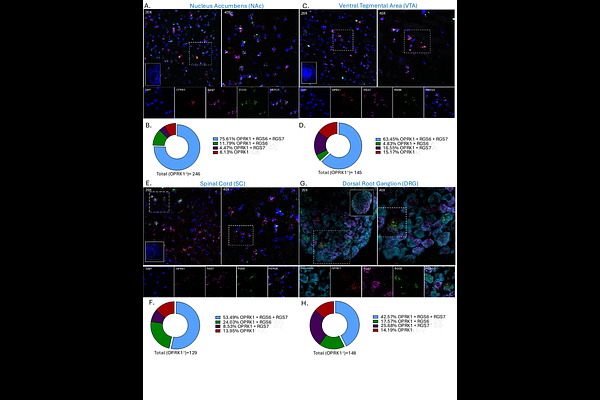
AbstractTargeting the kappa opioid receptor (KOR) system has emerged as a potential alternative to current analgesics, however, advancing the therapeutic development of KOR requires further elucidation of its intracellular signaling events and modulators. Among these intracellular modulators, Regulators of G protein signaling (RGS) proteins act as key modulators of GPCR signaling to shape nociceptive circuits and influence pain processing. Despite this, the molecular diversity of RGS proteins that shape KOR signaling and its behavioral consequences remains largely unexplored. Here we report that RGS6, a member of the R7 RGS family, is highly expressed in nociceptive areas and modulates multiple modalities of KOR-dependent anti-nociception and nocifensive behaviors. Using global single and double knockout mouse models we show that this anti-nociceptive phenotype was highly specific to RGS6 within the R7 RGS family. Further we demonstrate that the R7 RGS family displays a lack of functional redundancy in regulation of KOR signaling and behaviors. Using peripherally restricted KOR agonists, we found that KOR-RGS6 anti-nociceptive signaling displays sex differences in a site-specific manner, as females but not males displayed enhanced anti-nociceptive and blunted nocifensive behaviors. Our findings suggest that RGS6 is a highly specific modulator of KOR-dependent anti-nociceptive signaling and plays an essential role in modulating nociceptive circuits, potentially aiding in the development of novel analgesic drugs and therapeutics.